French Health Authority (HAS) recognizes medical use of EXCOR®
In the course of a human life the heart beats around three billion times. According to the European Society of Cardiology up to 10 million people in Europe suffer from cardiac insufficiency; approximately 500,000 new cases are diagnosed annually. Here an initial cardiac insufficiency can be treated very well with medication. However if the illness develops into a more advanced stage, a heart transplant is often the last resort.
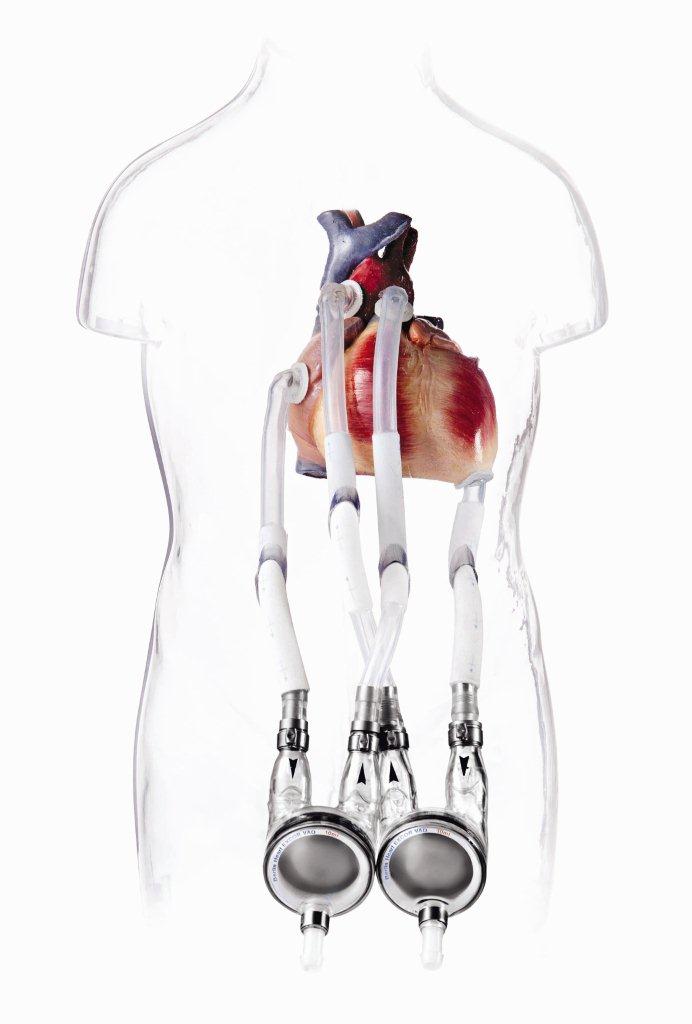
Because of the low availability of donor organs, the waiting periods are very long – often longer than a year. Those affected can only survive these waiting periods with a ventricular assist device. The EXCOR® System from the Berlin Heart company is such a system.
(firmenpresse) - In the course of a human life the heart beats around three billion times. According to the European Society of Cardiology up to 10 million people in Europe suffer from cardiac insufficiency; approximately 500,000 new cases are diagnosed annually. Here an initial cardiac insufficiency can be treated very well with medication. However if the illness develops into a more advanced stage, a heart transplant is often the last resort.
Because of the low availability of donor organs, the waiting periods are very long – often longer than a year. Those affected can only survive these waiting periods with a ventricular assist device. The EXCOR® System from the Berlin Heart company is such a system.
With this ventricular assist device all patients, from newborns to adults, can be optimally supported for single or double-sided heart failure.
To date over 3000 patients worldwide have profited from this therapy. Among them are also 50 children from France between the ages of three months and 16 years. Funds from the Program for Innovate and Expensive Therapies (STIC) have enabled this treatment, thus saving the small patients. After the French Health Authority assessed the ventricular assist device as especially valuable in treating children with severe heart failure, and because there is no alternative for the little patients, the therapy for children was added to the list of reimbursable products and therapies in December 2011. Now, in July 2012, the registration of the therapy for adults followed as well. Thus the proven therapy is available for the first time for all French patients.
Themen in dieser Pressemitteilung:
Unternehmensinformation / Kurzprofil:
About Berlin Heart
Berlin Heart GmbH is the only company worldwide which develops, manufactures and distributes both implantable ventricular assist devices and systems lying external to the body for patients with heart disease of all ages and body sizes. The systems INCOR®, EXCOR® Adult and EXCOR® Pediatric support short- to long-term pumping function of the heart and thus present a life-saving therapy possibility for the patient. Furthermore the user can access clinical and technical support around the clock. Berlin Heart is the market leader in Germany and Europe.
Contact:
Berlin Heart GmbH
Katharina Schubert
Marketing & PR
Wiesenweg 10
12247 Berlin
schubert(at)berlinheart.de
Contact:
Berlin Heart GmbH
Katharina Schubert
Marketing & PR
Wiesenweg 10
12247 Berlin
schubert(at)berlinheart.de
Datum: 13.09.2012 - 11:26 Uhr
Sprache: Deutsch
News-ID 182940
Anzahl Zeichen: 2344
contact information:
Contact person: Katharina Schubert
Town:
Berlin
Phone: 03081872650
Kategorie:
Medical Devices
Typ of Press Release: Unternehmensinformation
type of sending: Veröffentlichung
Date of sending: 13.09.2012
Diese Pressemitteilung wurde bisher 414 mal aufgerufen.
Die Pressemitteilung mit dem Titel:
"French Health Authority (HAS) recognizes medical use of EXCOR® "
steht unter der journalistisch-redaktionellen Verantwortung von
Berlin Heart GmbH (Nachricht senden)
Beachten Sie bitte die weiteren Informationen zum Haftungsauschluß (gemäß TMG - TeleMedianGesetz) und dem Datenschutz (gemäß der DSGVO).





